Digital Twins in Health: Beyond Marketing
Table of contents
Actualizado: 2026-05-03
Digital twins in health have spent years at conferences and in vendor marketing materials. After a long hype cycle, in 2024 there are concrete cases with measured results. This article separates what works from what remains vision: virtual patients, hospital optimisation, and the regulatory and technical obstacles nobody mentions in the brochure.
Key takeaways
- Operational twins (ER, ORs, logistics) are the most mature with the fastest ROI.
- Direct clinical twins (dosage, prognosis) are the most promising and require the most regulation.
- EU MDR classifies twins affecting diagnosis or treatment as medical software: years of validation required.
- GDPR and consent are real obstacles to sharing clinical data between institutions.
- The deterministic twin + LLM combination is the emerging trend for conversational interface and literature synthesis.
Three application layers
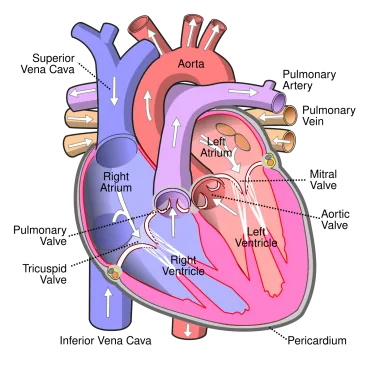
Patient twin: computational model of an individual — physiology, drug response, disease progression. Uses include treatment simulation before administration, clinical research with synthetic patients, and surgical planning (Siemens Healthineers, Dassault Systèmes Living Heart).
Hospital twin: operational model of a hospital — patient flows, resources, capacity. Measured results are the most solid: Basel Hospital’s ER twin reduced waiting times by 15%, NHS UK ambulance management pilots, GE Healthcare MRI uptime management. Typical ROI: 12-36 months for well-designed cases.
Device twin: predictive maintenance of MRI and CT scanners, remote calibration, staff training.
Real obstacles
Data: fragmentation between HIS, PACS, and LIS with different formats. GDPR makes inter-institutional data sharing complex. Legacy clinical data is dirty.
Regulation: EU MDR classifies as medical software (class IIa/IIb) any twin affecting diagnosis or treatment. Clinical validation is mandatory and can take years. Planning compliance from day one is not optional.
Clinical validation: a twin recommending chemotherapy dosage needs validation against real outcomes, ethical review, and trials if used as critical decision support. Errors have real consequences.
When adoption is worth it
Adopt now if: the hospital has IT capacity and a dedicated data science team, the use case is operational with clear ROI, or the organisation is pharma looking to reduce trial participants.
Wait if: the hospital is small without a dedicated team, the use case is high-risk direct clinical without proven validation, or EHR integration is immature.
Conclusion
Digital twins in health are exiting hype toward measurable applications. Operational twins are the most mature; direct clinical twins will take longer due to necessary validation. For hospitals wanting to start, operational focuses with existing data are the lowest-risk path. EU MDR regulation is a critical factor: plan compliance from the first sprint. Long-term, the combination of twins and generative AI promises genuine transformation of personalised medicine.